The standard of care for fit patients with unresectable, locally advanced (LA-) NSCLC is concurrent chemoradiation (CRT) followed by consolidation durvalumab. However, thoracic radiation (RT) for lung cancer carries a substantial risk of toxicity. The risk of acute and subacute pulmonary toxicity has long been recognized, but cardiac injury was traditionally believed to be a late toxicity, developing years after completion of RT. However, results from Radiation Therapy Oncology Group (RTOG)/NRG 0617, a phase III trial evaluating both RT dose escalation and the addition of cetuximab (C225), highlighted the effect of cardiac dose on mortality during the first 2 years following RT. Dose escalation to 74 Gy resulted in decrements in OS compared to standard doses of 60 Gy, while the addition of C225 failed to have any benefit whatsoever. Subsequent sub-group analyses showed a strong linear association between cardiac dose and survival, with volume of whole-heart receiving 5 Gy (V5) and 30 Gy (V30) highly statistically associated with OS.1
These observations led to subsequent studies that have attempted to confirm and refine this association. Several post-hoc analyses of large datasets have explored the association among cardiac dose, cardiac events, and OS. Wang et al.2 preformed a pooled analysis of 127 patients with LA-NSCLC treated with dose-escalated thoracic RT in six prospective trials. With a median follow-up of 8.8 years, researchers found that mean heart dose was associated with development of symptomatic cardiac events (i.e., pericardial effusion, acute coronary syndrome, pericarditis, significant arrhythmia, and heart failure), and it remained significant in a multivariable analysis that accounted for baseline risk. However, cardiac dose was not associated with OS.
Identifying Cause & Effect
Limiting cardiac dose remains challenging, given the many competing risks of lung, esophageal, and spinal cord toxicity, as well as the overriding need for tumor control. Several groups have explored whether dose to specific cardiac substructures predicts cardiac events or OS. A study by Wang et al.3 evaluated the previously described cohort 127 of patients from six prospective trials to determine the association between dose to cardiac substructures and cardiac events. They found that pericardial events were associated with whole-heart, right-atrial, and left-atrial dose, but not left-ventricular dose; ischemic events were associated with dose to the left ventricle and whole heart. Yegya-Raman et al.4 retrospectively analyzed a cohort of 140 patients treated with CRT for LA-NSCLC and analyzed development of symptomatic cardiac events and survival. They found that the dose to the whole-heart, ventricles, and left anterior descending artery were associated with subsequent development of congestive heart failure and acute coronary syndrome.
Several studies have also examined the association between pre-existing cardiac disease and cardiac RT risk. Dess et al.5 analyzed a cohort of 125 patients from four prospective multicenter trials who were treated with thoracic RT or CRT. On multivariable analysis, pre-existing cardiac disease was significantly associated with grade > 3 cardiac events (HR 2.96; 95% CI [1.04, 2.99]), as was mean heart dose Atkins et al.6 analyzed 748 patients treated with thoracic RT for LA-NSCLC and explored the association between cardiac dose, major adverse cardiac events, and all-cause mortality, adjusting for pre-existing coronary heart disease (CHD). They found that mean heart dose (stratified by ≤ 10 Gy vs. > 10 Gy) was associated with increased risk of all-cause mortality among patients without an existing history of CHD, but not among those with pre-existing CHD.
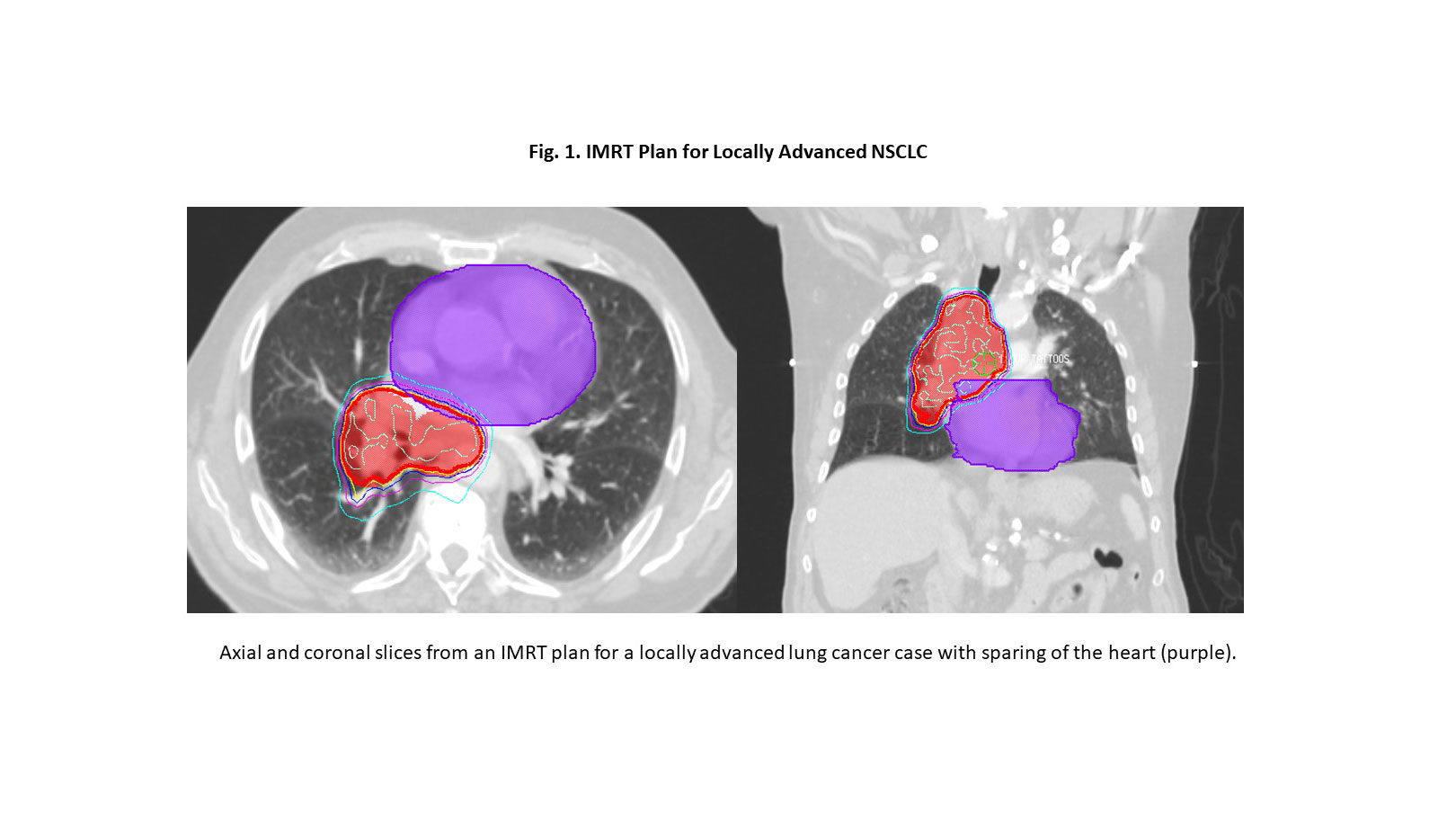
Advanced RT planning approaches offer the potential to reduce cardiac dose without compromising tumor coverage or increasing dose to the lungs. Intensity-modulated radiation (IMRT) and volume-modulated arc therapy (VMAT) allow conformal sculpting of photon RT to limit the dose to critical structures (Fig. 1). A secondary analysis of RTOG/NRG 0617 demonstrated that the use of IMRT was associated with lower cardiac doses compared to 3-D conformal RT.7 Proton therapy also holds potential promise for reducing cardiac dose and is being tested in the randomized phase III trial RTOG/NRG 1308.
In aggregate, most published studies suggest an association among cardiac dose, cardiac events, and survival following CRT for LA-NSCLC. However, a systematic review failed to identify consistent dose–volume parameters associated with OS, and the authors highlight the potential for both publication bias and multiplicity of testing, increasing the risk of type I errors.8 Additional well-designed studies with pre-determined endpoints and large datasets remain crucial in this space. Advanced RT technologies offer the potential to limit cardiac dose but require further prospective evaluation. Additionally, risk appears to increase in a linear fashion with dose, without clear “safe” dose constraints to guide clinical practice, particularly in the setting of a lethal disease. Current best practice remains full coverage of the tumor planning target volume to the prescription dose while using advanced technologies to minimize cardiac dose, as feasible.
References:
1. Bradley JD, Paulus R, Komaki R, et al. Standard-dose versus high-dose conformal radiotherapy with concurrent and consolidation carboplatin plus paclitaxel with or without cetuximab for patients with stage IIIA or IIIB non-small-cell lung cancer (RTOG 0617): a randomised, two-by-two factorial phase 3 study. Lancet Oncol. 2015;16(2):187-199.
2. Wang K, Eblan MJ, Deal AM, et al. Cardiac Toxicity After Radiotherapy for Stage III Non-Small-Cell Lung Cancer: Pooled Analysis of Dose-Escalation Trials Delivering 70 to 90 Gy. J Clin Oncol. 2017;35(13):1387-1394.
3. Wang K, Pearlstein KA, Patchett ND, et al. Heart dosimetric analysis of three types of cardiac toxicity in patients treated on dose-escalation trials for Stage III non-small-cell lung cancer. Radiother Oncol. 2017;125(2):293-300.
4. Yegya-Raman N, Wang K, Kim S, et al. Dosimetric Predictors of Symptomatic Cardiac Events After Conventional-Dose Chemoradiation Therapy for Inoperable NSCLC. J Thorac Oncol. 2018;13(10):1508-1518.
5. Dess RT, Sun Y, Matuszak MM, et al. Cardiac Events After Radiation Therapy: Combined Analysis of Prospective Multicenter Trials for Locally Advanced Non-Small-Cell Lung Cancer. J Clin Oncol. 2017;35(13):1395-1402.
6. Atkins KM, Rawal B, Chaunzwa TL, et al. Cardiac Radiation Dose, Cardiac Disease, and Mortality in Patients With Lung Cancer. J Am Coll Cardiol. 2019;73(23):2976-2987.
7. Chun SG, Hu C, Choy H, et al. Impact of Intensity-Modulated Radiation Therapy Technique for Locally Advanced Non-Small-Cell Lung Cancer: A Secondary Analysis of the NRG Oncology RTOG 0617 Randomized Clinical Trial. J Clin Oncol. 2017;35(1):56-62.
8. Zhang TW, Snir J, Boldt RG, et al. Is the Importance of Heart Dose Overstated in the Treatment of Non-Small Cell Lung Cancer? A Systematic Review of the Literature. Int J Radiat Oncol Biol Phys. 2019;104(3):582-589.