Primary results of the phase II Lung Cancer Mutation Consortium (LCMC) 3 clinical trial suggest that neoadjuvant atezolizumab may be a worthwhile treatment option for selected patients with resectable stage IB-IIIB NSCLC (Abstract PS01.05). Not only did 21% of patients lacking EGFR or ALK genetic aberrations attain a major pathologic response (MPR) at the time of resection, thus meeting the primary endpoint of the study, but 7% achieved a pathologic complete response (pCR). Moreover, 43% of 155 evaluable patients were downstaged followed neoadjuvant atezolizumab, whereas 19% were upstaged.
“Following neoadjuvant atezolizumab, resection was performed safely with low perioperative morbidity and mortality soon after completion of neoadjuvant therapy, infrequently outside of the protocol window, and with high complete resection rates,” added Jay M. Lee, MD, Chief of the Division of Thoracic Surgery at the Ronald Reagan UCLA Medical Center in Los Angeles, who presented the LCMC3 primary analysis findings during the Presidential Symposium.
Other neoadjuvant trials in NSCLC have evaluated immune checkpoint inhibitors (ICIs) prior to surgery, but typically have been much smaller studies either as ICI monotherapy or combination with platinum-based chemotherapy. LCMC3 is unique in that it is the largest trial of single-agent treatment with an ICI in the neoadjuvant setting, thus establishing a benchmark against which to compare the results of other preoperative ICI monotherapy or combination ICI and chemotherapy trials.
Study Details
LCMC3 featured an open-label, single-arm design (NCT02927301). A total of 181 patients with unselected resectable stage IB-IIIA and select IIIB NSCLC participated, all of whom received up to two cycles of neoadjuvant atezolizumab 1200 mg spaced 3 weeks apart (ie, on Days 1 and 22), followed by planned resection 8 to 28 days from the end of ICI (Days 30-50). Dr. Lee emphasized that the earlier and tighter 20-day protocol window for resection in LCMC3 differs vastly from historical neoadjuvant chemotherapy trials, which typically have a later and wider protocol window for surgery, whereby patients usually undergo resection after 21 days and up to 56 days from the end of preoperative chemotherapy.
Of the 181 patients included in the trial, 159 (88%) proceeded to surgery and did so within a median of 22 days (range: 11-74 days) from the last cycle of atezolizumab. Notably, 140 of the 159 patients (88%) who underwent disease resection did so within the Day 30-50 protocol window. Among the 19 patients who did not undergo surgical resection within the 20-day window, this was largely due to logistical reasons (n = 9) or other medical reasons (n = 6) and not due to treatment-related factors (n = 4).
In terms of the surgical procedure itself, minimally invasive surgery was initiated in 101 patients, with only 15 individuals (15%) converted to thoracotomy. This yielded a 54% rate of minimally invasive surgery using either robotic surgery of video-assisted thoracoscopic surgery and a 46% rate of thoracotomy. R0 resection was achieved in 92% of individuals, which is superior or comparable to historical neoadjuvant chemotherapy trials.
“Feasibility, on the whole, is acceptable, including the completion ratio of planned minimally invasive surgery. This fact suggests that the neoadjuvant therapy with ICI may not affect the difficulty of surgery,” remarked Shinichi Toyooka, Director of the Department of General Thoracic Surgery at Okayama University Hospital in Japan, who critiqued the LCMC3 findings.
Perioperative morbidity and mortality with atezolizumab were generally favorable. Remarkably, only 3% of patients experienced intraoperative bronchial or vascular complications, all of which were successfully repaired during the same operation. Patients were hospitalized for a median of 7.5 days (range: 2-68 days) following surgery, consistent with historical data. One patient died suddenly, not attributed to investigational therapy, within 2 weeks after surgery, yielding a 30-day postoperative mortality rate of 0.6%. Another individual died of pneumonitis within 2.5 months after surgery—an event considered related to atezolizumab—bringing the 90-day postoperative mortality rate to 1.3%, though still lower than the historical postoperative mortality rate of 2.2%.
Patients tolerated neoadjuvant atezolizumab fairly well. Prior to surgery, 6% of patients experienced grade ≥ 3 treatment-related adverse events and 2% experienced grade ≥ 3 immune-related adverse events, all of which were grade 3 in severity. After surgery, the rates of grade ≥ 3 treatment-related adverse events and immune-related adverse events increased to 13% and 8%, respectively.
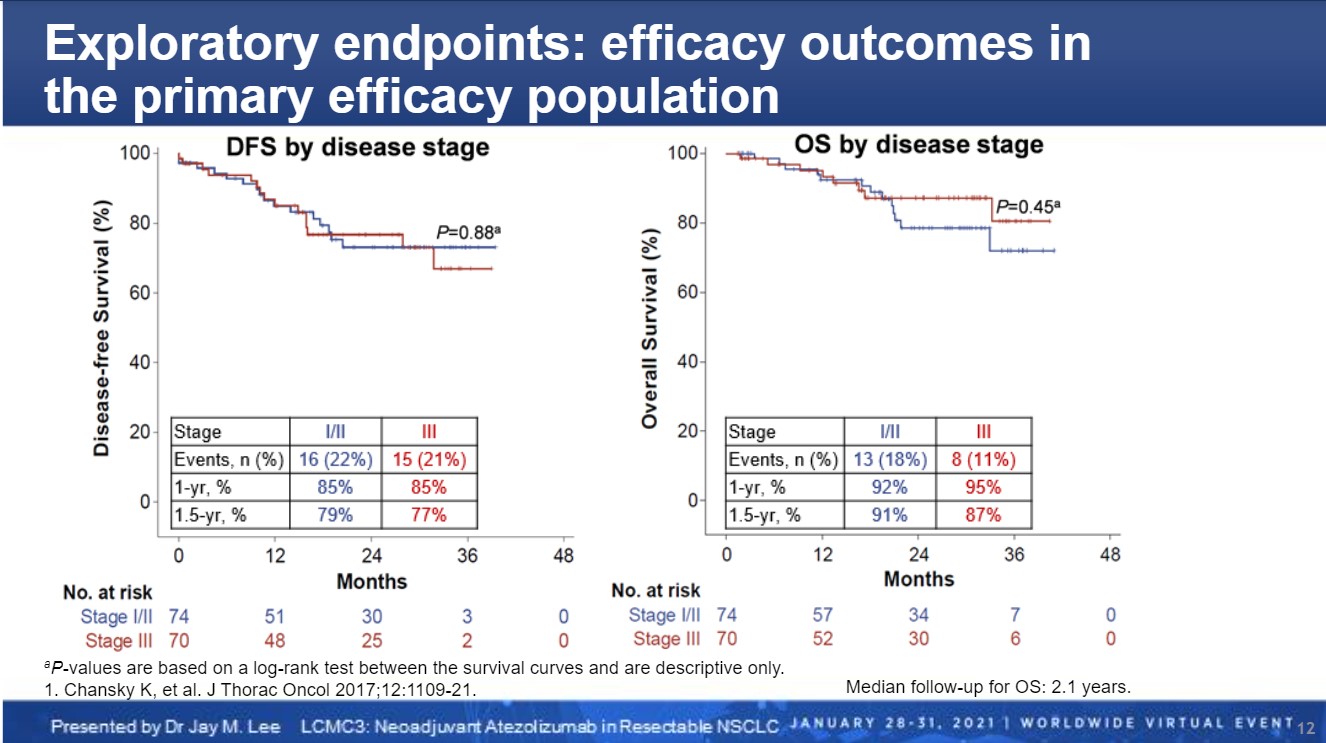
Based on a median follow-up of 2.1 years, clinical outcomes for the LCMC3 population are promising. For example, 79% of patients with stage I/II disease and 77% with stage III disease remain disease free at 1.5 years. Similarly, 91% and 87%, respectively, remain alive at 1.5 years.
“There is a suggestion that neoadjuvant atezolizumab has a survival advantage compared to historical information,” Dr. Lee remarked.
Given all the emerging data for ICI monotherapy and ICI-chemotherapy combinations in the neoadjuvant setting, the best approach to utilize in patients with resectable NSCLC remains to be determined.
Dr. Toyooka noted that although the LCMC3 results show nearly equivalent survival outcomes for patients with stage I/II disease versus those with stage III disease, he also emphasized that the data are still immature. “Thus, I think that single ICI as neoadjuvant therapy can be used for early-stage disease and patients with poor performance status. On the other hand, a combination of ICI and chemotherapy is suitable for advanced but resectable cases,” for example, based on supportive data from the NADIM clinical trial of neoadjuvant nivolumab plus platinum-based chemotherapy in patients with resectable stage IIIA NSCLC.1
Biologic specimens, including tumor tissue, normal lung tissue, lymph nodes, and blood were rigorously collected in LCMC3 to support translational studies. Analysis of these specimens may facilitate identification of predictive biomarkers that help to determine whether ICI monotherapy or combination therapy may be more appropriate in the neoadjuvant setting for each individual patient with NSCLC.
Reference
- Provencio M, Nadal E, Insa A, et al. Neoadjuvant chemotherapy and nivolumab in resectable non-small-cell lung cancer (NADIM): an open-label, multicentre, single-arm, phase 2 trial. Lancet Oncol. 2020;21(11):1413-1422.