The IASLC 's strategic initiative to advance the application of quantitative imaging for detection and management of early lung cancer especially through CT-based early lung cancer detection.
The Early Lung Imaging Confederation (ELIC): A Shared Network of Imaging Resources
This initiative is the Early Lung Imaging Confederation (ELIC) and it is an international alliance of collaborating individuals and institutions who share a vision to develop a globally distributed, privacy-secured, lung cancer imaging database and computational analysis environment. ELIC is designed to enable the analysis and study of extremely large collections of quality-controlled internationally assembled CT lung cancer images and associated biomedical data. A fundamental goal of ELIC is to support the development of deep learning methods or artificial intelligence approaches to detect early lung cancer and other diseases using thoracic CT images, characterize small pulmonary nodules and surrounding structures, and to measure responsiveness to therapeutic interventions.
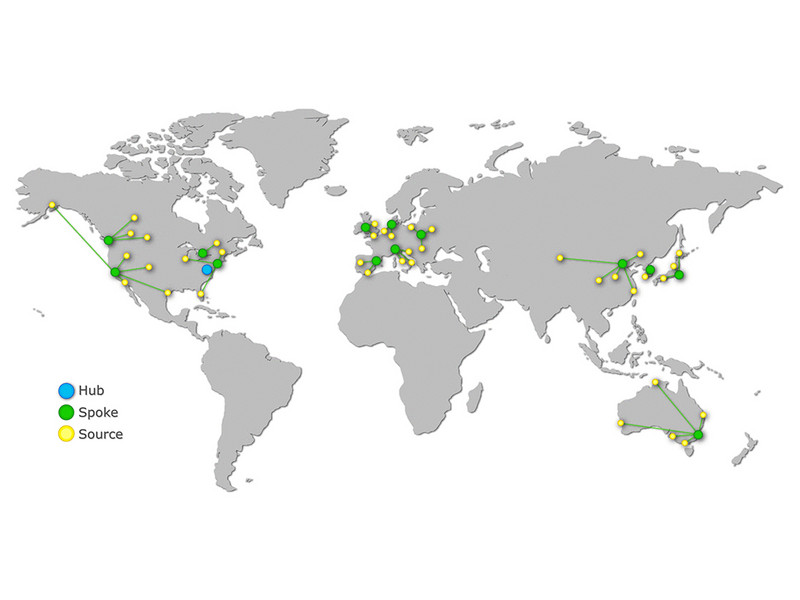
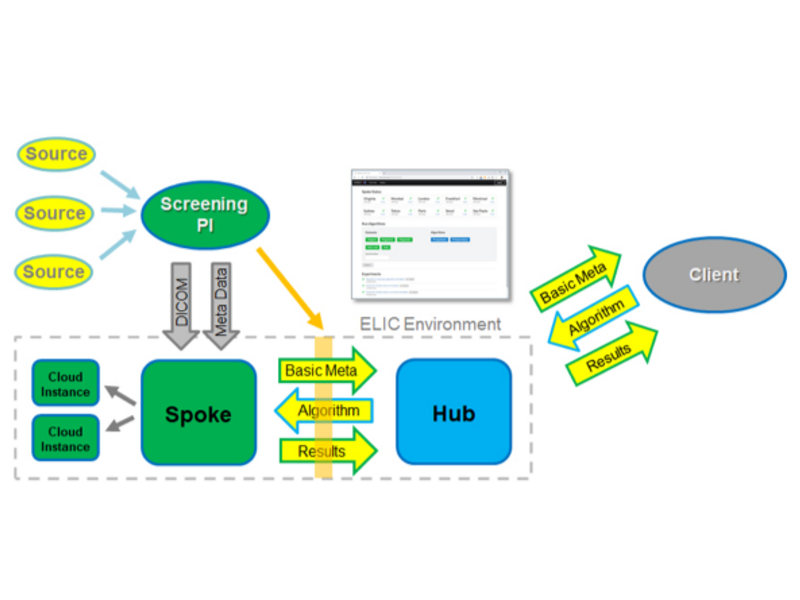
The ELIC infrastructure is being developed using a Hub and Spoke model and leveraging cloud computing resources and will allow clinical research groups (Spokes) to securely make their locally stored, de-identified lung cancer imaging collections available for computational analysis by other research groups (Clients), all coordinated by a central ELIC managed server (Hub).
Phase I
This initial proof of concept project demonstrated the potential of the ELIC Hub and Spoke Environment (H&SE) to provide a useful resource for global quantitative lung imaging studies. For this initial phase global cloud-computing resources populated with over 100 publicly available and de-identified CT lung cancer screening image datasets were used. A central hub website simultaneously distributed two open source quantitative lung measurement algorithm requests to 10 globally distributed virtual spoke sites that contained a copy of the publicly available CT lung cancer screening image datasets.
Phase II
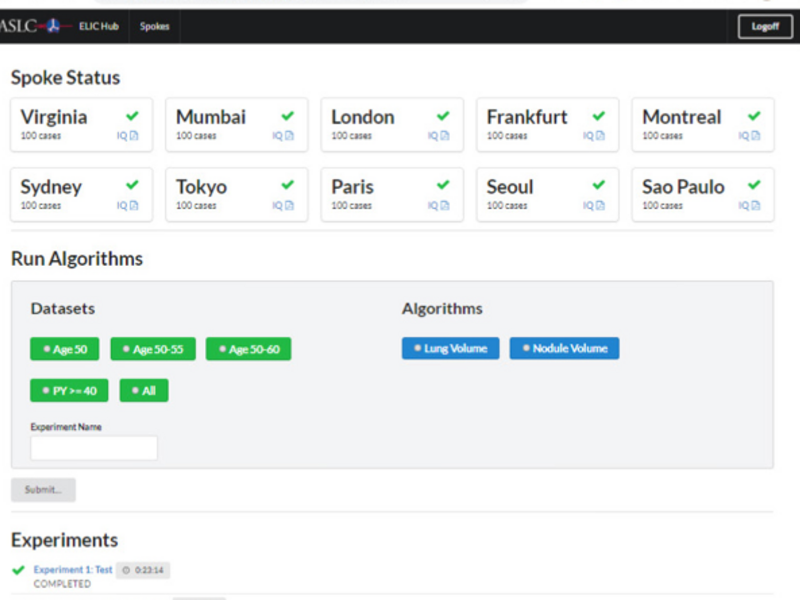
The ELIC H&SE infrastructure is undergoing further development to bring it from a proof-of-concept pilot to a functional globally distributed database and computational environment capable of performing useful quantitative lung cancer imaging studies. References to tools and resources for performing data de-identification are being added to support research groups that will be uploading lung imaging datasets and metadata into the ELIC H&SE. A technical manual has been created that gives spoke investigators technical specifications and methods for uploading images and metadata as well as guidance on how clients can participate in the ELIC H&SE. A separate document gives guidance on the process of installing and using Clinical Trial Processor (CTP), an open source DICOM de-identification tool. The initial spokes, six have been identified, will 1) identify the necessary images and data; 100 cases with 2 images per case (25 with cancer, 25 no cancer, no nodules, and 50 with nodules but no cancer detected), pull the images and data from their current database, de-identify the data and format it for spoke uploading, upload the data into the spokes, and monitor the spoke. After the sites upload their initial 100 each cases of images and data, multiple quantitative lung analysis algorithms, from Accumetra/ELIC or the contributing institutions, will be run on the globally distributed datasets within the spokes.
The IASLC is grateful for the participation of sites and investigators worldwide who are contributing their time and their data toward advancing this project.
The IASLC would like to thank AstraZeneca for their support of Phase II.
Phase III
Phase 3 will scale up the ELIC H&SE and expand the number of spokes by a factor of 2 or 3 and increase the number of images by a factor of 100 or 1000. Deep learning and artificial intelligence approaches will be applied to the large data set to improve the accuracy of malignancy determination of pulmonary nodules, determine other imaging biomarkers, improve the work stream of radiologists, and create better risk criteria for defining who should be screened in the future.