The International Association for the Study of Lung Cancer
The IASLC is a global multidisciplinary organization dedicated to eradication of all forms of lung cancer. From provision of educational events around the world and virtually to research projects and publications that advance the science of lung cancer, the IASLC's members—consisting of medical, surgical, and radiation oncologists, as well as other thoracic oncology specialists such as nurses, basic scientists, pathologists, radiologists, pulmonologists, statisticians, patient research advocates, patients, and their caregivers—are raising the bar for care of patients with lung cancer.
ILCN In Focus

Thoracic Circle is the IASLC's official online community platform designed to foster engagement, encourage knowledge sharing, and facilitate collaboration among active IASLC members. This dynamic virtual environment is a perfect place to share resources, discuss complex cases, exchange insights across specialties, network, and stay informed on the latest advances in thoracic oncology.
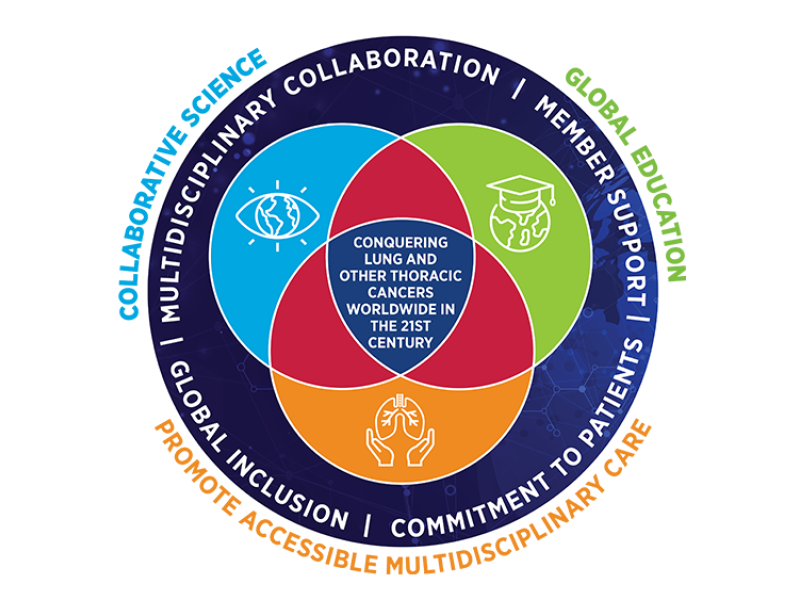
IASLC's Visionary Strategic Plan for the Next Five Years
In the ever-evolving landscape of lung cancer research and patient care, we have embarked on a transformative journey by unveiling a new strategic plan that redefines its mission and objectives for the next five years.

IASLC's STARS Program
The STARS Program (Supportive Training for Advocates on Research & Science) was formed in 2019 to address this need. A collaboration composed of patient and professional lung cancer research advocates and lung cancer advocacy organizations designed STARS to help patient advocates evolve into patient research advocates (PRAs), increasing the number of PRAs equipped to provide accurate scientific translation in their online or real-life groups for people living with lung cancer, and enabling more people to provide the patient perspective for lung cancer research and policy.

IASLC's Atlas of Molecular Testing for Targeted Therapy in Lung Cancer
The IASLC’s latest Atlas provides a comprehensive overview of all current and emerging molecular targets, specimen acquisition, available assays, and reporting and interpretation of molecular testing results from both a diagnostic and clinical perspective.

IASLC's Corporate Roundtable
Born from feedback received during meetings with our Corporate Partners in 2022, the IASLC Partners for Thoracic Cancer Care brings IASLC leadership and Roundtable Member Representatives together to collaborate on solutions to big-picture challenges facing those working to conquer lung cancer and other thoracic malignancies.
Upcoming Meetings & Webinars

Journal of Thoracic Oncology & JTO Clinical and Research Reports
Lung and thoracic specialists count on the IASLC’s journals to provide perspective on innovation in prevention, detention, diagnosis and cutting-edge treatments. As a leading medical journal, JTO focuses on lung and thoracic cancers and is dedicated to presenting original research reviews and opinions from a wide range of disciplines within the medical community. JTO CRR, the official open access journal of the IASLC, complements the JTO by offering authors a gold open access publication option.








